|
12/30/2023 0 Comments Generic vsepr shapes practice 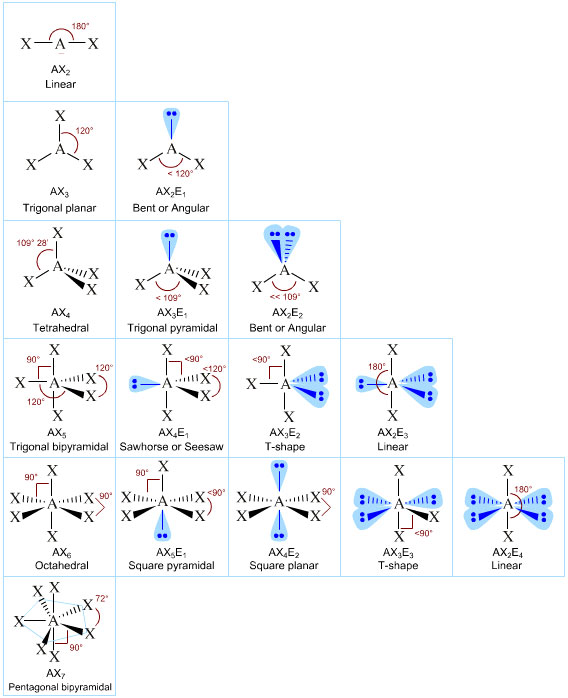
Caused by polar bonds and asymmetric geometry. They have a positive and negative region of the molecule. Determine both molecular geometry and shape. In a complete analysis of the geometry of a molecule it would be necessary to consider such factors as nuclear-nuclear interactions, nuclear-electron interactions, and electron-electron interactions. Terms in this set (25) Electron pairs repel and molecules adjust their shapes so that valence electron pairs and bonds are as far apart as possible. Worksheet 3- Complicated VSEPR Shapes.doc - Course Hero VSEPR practice problems Draw the 2-D LEWIS structure below the molecular formula. 2 A multiple bond (double bond or triple bond) counts as one bond in the VSEPR model. Phet Molecular Shapes Worksheet Answers : Molecule Polarity Phet Lab berasketaann. 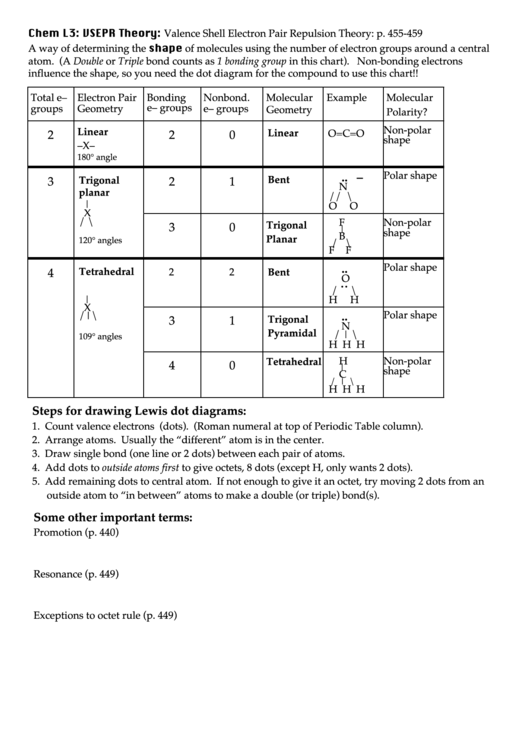
VSEPR Shapes Practice ENDORSED Post by Luis Torres 1C » Fri 3:20 am. This has the angles, bonds, and shapes of different molecules Learn with flashcards, games, and more for free. Phet Molecular Geometry Part 1 Generic Molecules Fill in the chart. Luis Torres 1C Posts: 30 Joined: Fri 6:05 pm. Despite this, the correct geometry is nearly always predicted, and the exceptions are often rather special cases. VSEPR Shapes Practice ENDORSED (Polar molecules, Non-polar molecules, etc.) Moderators: ChemMod, ChemAdmin. Organic molecules are treated just as successfully as inorganic molecules.Īpplication of the VSEPR method requires some simplifying assumptions about the nature of the bonding. Valence shell electron pair repulsion theory, VSEPR, is a super-simple technique for predicting the shape or geometry of atomic centres in small molecules and molecular ions: Crucially, atomic centres with VSEPR determined geometry can be joined together into molecular entities like cyclohexane and glucose: This molecular building-block logic. It is a remarkably simple device that utilizes a simple set of electron accounting rules in order to predict the shape of, in particular, main group compounds. For main group compounds, the VSEPR method is such a predictive tool and unsurpassed as a handy predictive method. 2 H 2 Worksheet 13 - Molecular Shapes The shapes of molecules can be predicted from their Lewis structures by using the VSEPR (Valence Shell Electron Pair Repulsion) model, which states that electron pairs around a central atoms will assume a geometry that keeps them as far apart from each other as possible. 
It is also desirable to have a simple method to predict the geometries of compounds. It is very important to know the shape of a molecule if one is to understand its reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
